Are There Other Neurotransmitters Involved
While dopamine receives a large amount of attention when looking at schizophrenia symptoms, its not the only neurotransmitter researchers are eyeing with this disorder.
Of the over 40 different neurotransmitters studied by researchers today, there are four others that are believed to play a role in schizophrenia — glutamate, GABA, serotonin, and acetylcholine.
Lets take a closer look at these neurotransmitters and what theyre responsible for:
New Evidence And The Rationale For Version Iii
Much has changed since version II. There have been more than 6700 articles and 181000 citations to the topic of dopamine and schizophrenia since 1991. It is not possible to provide a comprehensive review of all the new findings since then, much less try to weave them into a coherent hypothesis. So, the focus of our effort is to identify the 5 most critical streams of new evidence, briefly summarize what we see as the key findings from these, and use them to develop the most parsimonious understanding of the role of dopamine in schizophreniaversion III.
How Does Dopamine Affect Schizophrenia
You might be wondering, What is the role of dopamine in schizophrenia? and where do schizophrenia and dopamine start to overlap.
Researchers have identified dopamine as one of the major neurotransmitters involved in schizophrenia symptoms. Over time, theyve discovered that the symptoms are largely caused by high levels of dopamine in the brain.
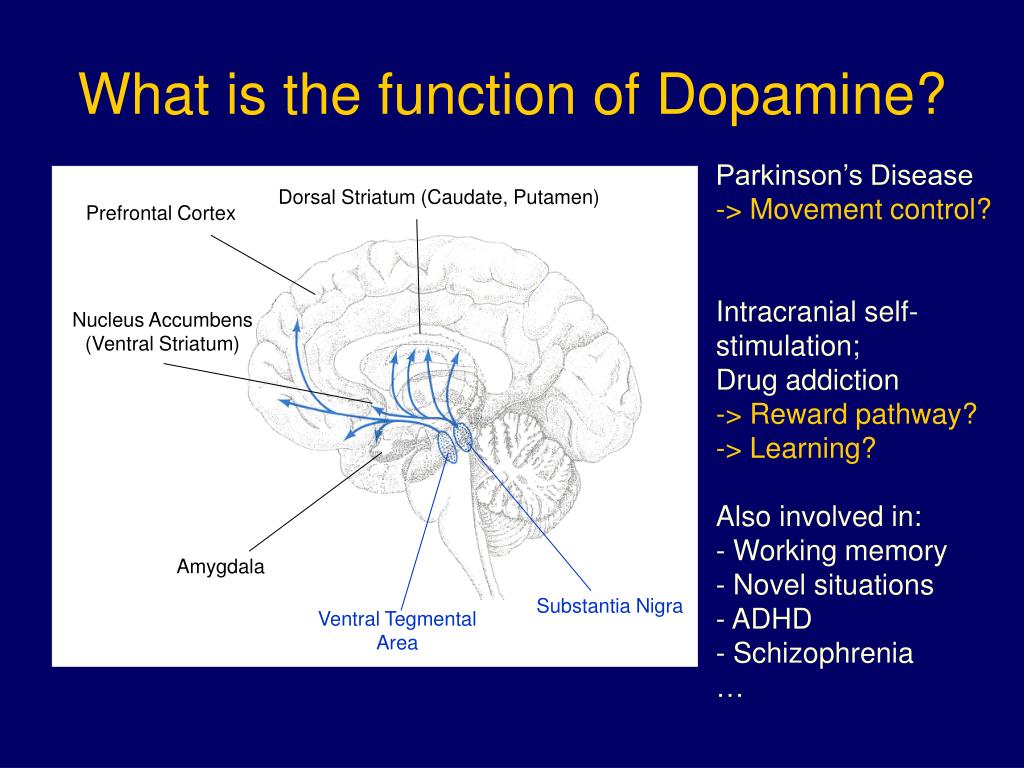
In fact, there are four major dopamine pathways in the brain that play a role in schizophrenia, and theyre often the main pathways targeted by antipsychotics prescribed by your doctor. Lets take a closer look at these four dopamine pathways and where theyre located in the brain:
These four dopamine pathways play a major role in understanding where positive and negative originate from. Medication prescribed by your doctor is designed to bind to dopamine receptors in these pathways.
Also Check: What Is The Difference Between Sadness And Depression
Effects Of Antipsychotics On Positive And Negative Symptoms
As discussed previously, schizophrenia involves impaired Go learning and blunted PE signaling, which may relate to negative symptoms, and antipsychotics may aggravate these reinforcement-learning deficits and some negative symptoms. Indeed, antipsychotics, administered chronically, reduce dopamine-neuron firing , so they may blunt adaptive dopamine transients, in addition to blunting their postsynaptic effects through D2 blockade. The consequent aggravation in reinforcement-learning deficits and some negative symptoms may help explain the poor adherence to antipsychotic treatment. Blunting of dopamine transients, however, may be precisely what improves positive symptomsalbeit by reducing spontaneous transients.
Striatal D2 Receptor Density And Blockade In Treatment
As already mentioned above, the blockade of a proportion of D2 receptors is not a working antipsychotic mechanism for a significant number of patients with schizophrenia . In fact, roughly one-third of individuals with schizophrenia are resistant to treatment with first-line antipsychotics despite sufficient D2 receptor occupancy . Clozapine, which works at a relatively low striatal D2 receptor occupancy , is the most effective antipsychotic in the majority of patients refractory to other antipsychotic medications . If we hypothetically accept the suggestion that this outcome is not attributable to D2 receptor binding kinetics , we begin to consider other dopaminergic mechanism that may account for this apparent discrepancy. A growing literature supports the idea that additional dopaminergic mechanisms may underlie therapeutic efficacy of antipsychotic drugs . Some patients who respond to first-line antipsychotic treatment experience diminished treatment efficacy over time , which can lead to treatment non-compliance and relapse . Diminished antipsychotic efficacy may also occur despite stable D2 receptor occupancy . These dynamics are depicted in Figure 1. The opposite has also been observed with long-term antipsychotic efficacy occurring despite decreasing D2 receptor occupancy .
You May Like: How To Help With Panic Attacks Anxiety
The Role Of Dat In Antipsychotic
Figure 4 Representation of the pharmacological mechanism underlying the absence of therapeutic response in antipsychotic-resistant schizophrenia based on our model. Aging and/or addictive drugs consumed before antipsychotic treatment begins lead to reduced expression of the dopamine transporter , D2 autoreceptors, and tyrosine hydroxylase , as these proteins appear to be co-regulated, at least in rodents. During environmentally evoked phasic dopamine release, impaired capacity for autoinhibition results from low levels of DAT and D2 autoreceptors. The resulting post-synaptic stimulation contributes to psychosis despite a significant blockade of D2 receptors by antipsychotic drugs .
What If I Have Too Little Or Too Much Dopamine
Dopamine dysregulation could mean that the brain is producing too little or too much dopamine. Low dopamine, or dopamine deficiency, can be caused by a variety of factors, including conditions such as Parkinsons disease, schizophrenia, and depression. Drug and sugar addiction have also been found to cause dopamine deficiency over time. Some low dopamine symptoms include fatigue, moodiness, dysphoria, physical pain, and changes in weight, sex drive, and ability to focus. As many other conditions share these symptoms, its important to consult your health care provider if youre experiencing these symptoms.
Its also possible to have too much dopamine. Effects of overly high dopamine levels include high libido, anxiety, difficulty sleeping, increased energy, mania, stress, and improved ability to focus and learn, among others. When certain parts of the brain are exposed to too much dopamine, for instance right after an individual takes illicit drugs, other behaviors may be present. These can include aggression, hallucinations, twitching, nausea and/or vomiting, and depression.
Recommended Reading: Can You Have A Panic Attack While High
Dopamine Cannabinoids And Schizophrenia
Owing to the historical prominence and wide familiarity of the dopamine hypothesis of schizophrenia, a natural question to ask is whether the psychotomimetic effects of cannabis are mediated by dopamine . Dopamine response is clearly blunted among chronic cannabis users, showing a dose-dependent effect . However, the degree of this effect is not correlated with the level of psychotic symptoms in these users , and the magnitude of the dopamine release also does not correlate with the degree of psychotic symptoms that are acutely produced by cannabinoids . These findings suggest that the effects of cannabis on psychosis are not explained by these alterations in the dopaminergic system. The modern dopaminergic hypothesis of schizophrenia is much more complex than hypothesizing cortical hypofunctioning and mesolimbic hyperfunctioning, and readers wanting a more in-depth analysis of the dopamine hypothesis of schizophrenia are directed to recent reviews on the subject .
Neural Circuits And Dopamine
The evidence discussed above suggests that, while the dopamine hypothesis can account for the positive symptoms of psychosis, it is less clear whether it can fully account for negative and cognitive symptoms. Similarly, while glutamatergic models of psychosis are able to replicate a wide range of symptoms of psychosis, they do not directly account for the finding of increased presynaptic striatal dopamine function, nor the clinical effectiveness of dopamine antagonists. This suggests that dysfunction in both systems contributes to the pathophysiology of schizophrenia, and highlights the need to understand how these two systems may interact.
Much research has investigated dopamine-glutamate relationships in humans using pharmacological challenges. Amphetamine administration has been shown to increase cortical glutamate levels, as measured using 1H-MRS, but dopamine antagonists do not have consistent effects on glutamate levels as measured using 1H-MRS. Several, but not all, PET studies have found that ketamine administration is associated with striatal dopamine release. While glutamatergic dysfunction may encourage dopaminergic disinhibition, it is clear that this is not the only route to symptoms, given that dopamine antagonists do not entirely ameliorate the effects of NMDA antagonists.
Also Check: What Is The Most Common Phobia In The World
Drug Addiction In Schizophrenia
Epidemiological studies report that nearly half of patients with schizophrenia also suffer from drug addiction . This is about four times more prevalent than in the general population . If we consider that the recreational consumption of addictive drugs is common in the general population , but only a small proportion of individuals exposed to drugs of abuse become drug addicted and that this happens about four times more often in patients with schizophrenia, then it is possible that many of the remaining 50% of patients with schizophrenia without formal diagnosis for drug addiction likely consume at least some class of addictive drugs as well. The most commonly consumed drugs in patients with schizophrenia include alcohol, psychostimulants, cannabis, and tobacco . It has been suggested that patients with schizophrenia may use illicit substances to self-medicate their symptoms as well as the side effects of antipsychotic medications , as self-medication with addictive drugs is indeed common in patients with mental illness .
Indirect Evidence For Dopamine Dysfunction In Schizophrenia
Animal models
Rodent models of schizophrenia are useful for investigating molecular mechanisms that may be of pathophysiological relevance, and for testing novel therapeutic interventions.
One well characterized model of dopaminergic hyperactivity involves administering repeated doses of amphetamine. This has been shown to induce events that are also observed in individuals with schizophrenia, such as reduced prepulse inhibition, stereotyped behaviours, and impaired cognitive flexibility and attention. Given that amphetamine results in dopamine release, and that the above effects can be ameliorated with the administration of dopamine antagonists, this provides indirect evidence for a role of dopamine in behaviour thought to be a proxy for psychotic symptoms.
Another example is that of mice genetically modified to overexpress dopamine D2 receptors in the striatum, which also display a wide range of schizophrenia-like behaviours. Similarly, transgenic insertion of tyrosine hydroxylase and guanosine triphosphate cyclohydrase 1 into the substantia nigra in early adolescence increases dopamine synthesis capacity, and has been associated with a schizophrenia-like behavioural phenotype.
In summary, multiple methods have been used to induce increased striatal dopamine signalling in animal models, and these consistently produce behaviours analogous to those observed in individuals with schizophrenia.
Cerebrospinal fluid and post-mortem studies
Summary of indirect findings
Read Also: Is Sleeping A Sign Of Depression
Are Schizophrenia Dopamine Levels High Or Low
Reviewed by Heather Cashell, LCSW
Schizophrenia is a serious and chronic brain disorder that is believed to affect over 20 million people all over the world. The symptoms are generally characterized by hallucinations and delusions, but people living with schizophrenia experience a wide variety of symptoms each day.
For example, individuals with schizophrenia could experience abnormal thinking, unwanted behaviors, a distorted view of whats real, changes in the five senses , confusion, loss of motivation, and even difficulty speaking.
Genetics and the way someone is raised are certainly believed to play a role in individuals developing schizophrenia. With that being said, its the impact dopamine, and other neurotransmitters have on the brains function that is at the heart of most research today. Thats where the connection between dopamine and schizophrenia begins.
So, what is dopamine?
Dopamine is a neurotransmitter found in the brain. Many people refer to it as the pleasure neurotransmitter because it plays a large role in feeling pleasure. In fact, dopamine is the chemical our brain releases when we do something we enjoy — it makes us feel happy.
Once produced, dopamine wont enter the bloodstream because it cant cross the blood-brain barrier. Instead, it travels through the neural pathways that make up the brain. These little roadways throughout the brain are what help different regions of the brain communicate with each other.
The Role Of Dopamine In Schizophrenia From A Neurobiological And Evolutionary Perspective: Old Fashioned But Still In Vogue

- 1Department of Forensic Medicine, Medical University of Gdask, Gdask, Poland
- 2School of Medical Sciences, The University of Adelaide, Adelaide, SA, Australia
- 3Centre for Evolutionary Medicine, University of Zurich, Zurich, Switzerland
- 4Department of Psychiatry and Psychotherapy, Ruhr University Bochum, Bochum, Germany
- 5Department of Psychiatry, Otto-von-Guericke-University of Magdeburg, Magdeburg, Germany
- 6Department of Zoology, Institute of Biology, Otto-von-Guericke-University of Magdeburg, Magdeburg, Germany
- 7Biological Anthropology and Comparative Anatomy Research Unit, School of Biomedical Sciences, The University of Adelaide, Adelaide, SA, Australia
Recommended Reading: How To Stop Separation Anxiety In Puppies
Evidence Against The Dopamine Hypothesis
Further experiments, conducted as new methods were developed challenged the view that the amount of dopamine blocking was correlated with clinical benefit. These studies showed that some patients had over 90% of their D2 receptors blocked by antipsychotic drugs, but showed little reduction in their psychoses. This primarily occurs in patients who have had the psychosis for ten to thirty years. At least 90-95% of first-episode patients, however, respond to antipsychotics at low doses and do so with D2 occupancy of 60-70%. The antipsychotic aripiprazole occupies over 90% of D2 receptors, but this drug is both an agonist and an antagonist at D2 receptors.
Furthermore, although dopamine-inhibiting medications modify dopamine levels within minutes, the associated improvement in patient symptoms is usually not visible for at least several days, suggesting that dopamine may be indirectly responsible for the illness.
The excitatory neurotransmitter glutamate is now also thought to be associated with schizophrenia. Phencyclidine and ketamine, both of which block glutamate receptors, are known to cause psychosis at least somewhat resembling schizophrenia, further suggesting that psychosis and perhaps schizophrenia cannot fully be explained in terms of dopamine function, but may also involve other neurotransmitters.
What Are The Symptoms Ofschizophrenia
AmericanPsychiatric AssociationDiagnostic and StatisticalManual of Mental Disorders, Fifth Edition
When people show any of these five symptoms, they are considered to be inthe “active phase” of the disorder. Oftenpeople with schizophrenia have milder symptoms before and after the active phase.
There are three basic types of schizophrenia. All people who haveschizophrenia have lost touch with reality. The three main types ofschizophrenia are:
Also Check: What Are The Best Treatments For Ptsd
The Role Of Dopamine In The Body
Dopamine has a direct impact on many neurological, cognitive, and behavioral functions within the body, says Dr. Giordano, including:
- Movement
- Difficulty sleeping
- Hallucinations
While high levels of dopamine can increase your concentration, your energy, your sex drive, and your ability to focus, it can also lead to competitive, aggressive behavior and cause symptoms including anxiety, trouble sleeping, and stress.
The Dopamine Hypothesis: Version Ii
In 1991, Davis et al published a landmark article describing what they called a modified dopamine hypothesis of schizophrenia that reconceptualized the dopamine hypothesis in the light of the findings available at the time. The main advance was the addition of regional specificity into the hypothesis to account for the available postmortem and metabolite findings, imaging data, and new insights from animal studies into cortical-subcortical interactions. It was clear by this stage that dopamine metabolites were not universally elevated in the cerebrospinal fluid or serum of patients with schizophrenia. Also the focus on D2 receptors was brought into question by findings showing that clozapine had superior efficacy for patients who were refractory to other antipsychotic drugs despite having rather low affinity for and occupancy at D2 receptors. Furthermore, the postmortem studies of D2 receptors in schizophrenia could not exclude the confounds of previous antipsychotic treatment, and the early positron emission tomography studies of D2/3 receptors in drug-naive patients showed conflicting results.
Recommended Reading: How Many Years Does Ptsd Last For
Disturbances In Reinforcement Learning And Pe Signaling
Impaired Go Learning and Blunted PE Signaling in Medicated Patients
Medicated schizophrenia patients exhibit impaired Go learning but preserved NoGo learning . Further supporting an impairment in Go learning, medicated patients fail to learn to speed up for cues for which faster responses give greater rewards . The impairment in Go learning correlates with negative symptoms , which seems intuitive: impaired learning from rewards with preserved learning from punishments could produce avolition . Consistent with the impairment in Go learning, medicated patients show blunted neural responses for positive PEs in the striatum, midbrain, and other limbic regions , which correlate with negative symptoms .
Consistent with spared NoGo learning, medicated patients show normal activity for negative PEs induced by reward omission and, in extrastriatal areas, even show increased activation for losses . However, medicated patients show reduced aversive Pavlovian conditioning and blunted activity for PEs elicited by aversive stimuli findings that may reflect the possible involvement in aversive conditioning of phasic responses in a subset of dopamine neurons .
Impaired Go Learning and Blunted PE Signaling Induced by Antipsychotics
Impaired Go Learning and Blunted PE Signaling in Unmedicated Patients
Relation to Dopamine Function
Dopamine Dysfunction In Schizophrenia
10.1 Dopamine Dysfunction in Schizophrenia; 10.2 Neuropharmacological Profiles of Antipsychotic Drugs; 10.3 How Antipsychotics Work: Linking Receptors to Response; 10.4 Dopamine Dysfunction in Schizophrenia: From Genetic Susceptibility to Cognitive Impairment; 10.5 The Role of Dopamine in the Pathophysiology and Treatment of Major Depressive …
- Estimated Reading Time: 2 mins
Read Also: Can Dramamine Help With Anxiety
Dopaminergic Features Of Psychosis In Schizophrenia
In healthy individuals, dopamine stimulants such as amphetamine can induce psychotic symptoms, and people with schizophrenia are more sensitive to these effects, . Studies using positron emission tomography imaging have shown patients with schizophrenia show increases in subcortical synaptic dopamine content, , abnormally high dopamine release after amphetamine treatment,,,,, and increased basal dopamine synthesis capacity ,, compared with healthy controls. Increased subcortical dopamine synthesis and release capacity are strongly associated with positive symptoms in patients, , and increased subcortical synaptic dopamine content is predictive of a positive treatment response. It was widely anticipated that the limbic striatum would be confirmed as the subdivision where these alterations in dopamine function would be localised in patients. The basis for this prediction was the belief that reward systems were aberrant in schizophrenia. However, as PET imaging resolution improved it was found that increases in synaptic dopamine content, and synthesis capacity were localised, or more pronounced, in the associative striatum . Furthermore, alterations in dopamine function within the associative striatum likely contribute to the misappropriate attribution of salience to certain stimuli, a key aspect of delusions and psychosis.
Environmental Risk Factors For Schizophrenia

A large number of disparate environmental factors clearly contribute to the risk for schizophrenia, yet many hypotheses of schizophrenia, including previous versions of the dopamine hypothesis, make no allowance for them. Markers of social adversity such as migration, unemployment, urban upbringing, lack of close friends, and childhood abuse are all associated with a well-established increased risk for schizophrenia that cannot readily be explained by genetic factors alone. These factors either directly index social isolation/subordination or are linked to these experiences. Studies in animals of social isolations and subordination, find that these factors lead to dopaminergic overactivity.
Don’t Miss: Is Insomnia A Symptom Of Depression
Natural Ways To Balance Dopamine Levels
Dopamine levels are difficult to monitor since they occur in the brain, but there are ways to balance your dopamine levels without medication. The best way to balance your dopamine levels is to focus on healthy habits.
If youre over indulging in certain dopamine-producing activities like sex, technology, or gambling, then youll want to take intentional breaks, but if youre having trouble concentrating, feeling unmotivated or tired, then youll want to increase your dopamine production.
Modelling Psychosis: The Use Of Animal Models
Potentially, the most useful avenue for animal models to assist in schizophrenia research will be identifying convergent aetiological pathways. Understanding which neurotransmitter systems and brain regions are most involved may help to identify the core neurobiological features of schizophrenia. For example, changes in dopaminergic systems are observed in animal models after manipulation of factors based on schizophrenia epidemiology, , genetics, pharmacology and related hypotheses. These include changes in early dopamine specification factors, , sensitivities to psychostimulants,,, and alterations in dopamine neurochemistry,,, . Evidence of subcortical dopaminergic hyperactivity or sensitivity in animal models is proposed to represent the face validity for psychosis in patients. The most commonly used behavioural assessments of positive symptoms in animal models include enhanced amphetamine-induced locomotion and deficits in prepulse inhibition . These tests are widely used because they are relatively simple to perform. However, we propose that given current knowledge of the neurobiology in schizophrenia, they have outlived their usefulness as measures of positive symptoms.
Also Check: How Do You Get Paranoid Schizophrenia
Dopamine Autoinhibition As A Feature Of Antipsychotic Responsiveness
This autoinhibition might be mediated by the D2S isoform since the two splice variants have distinct functions and are unevenly distributed within the striatonigral dopaminergic circuitry . Furthermore, antipsychotics appear to preferentially bind dopamine receptors in the striatum , a brain structure with predominant expression of D2L as discussed above, and dopamine exhibits higher binding affinity for D2S in transgenic mice and in cell culture . Together these data suggest that therapeutic doses of antipsychotics in the brain cause a functional segregation of D2S and D2L, which based on the data available until now could overlap with a functional segregation of pre- and post-synaptic D2 receptors . It should be noted that both isoforms are expressed in pre- and post-synaptic neurons and the functional segregation might also occur within the same cells . In support of this theory are studies with human schizophrenia patients demonstrating selective reduction in expression of D2S mRNA , potentially indicative of a desensitization of the short isoform in response to increased dopamine activity on this receptor. On the other hand, postmortem studies also show that D2L mRNA is upregulated in patients with schizophrenia , which may indicate an adaptive response to chronic blockade .
Hypofunctional Dopamine Uptake And Antipsychotic Treatment
- 1Department of Neuroscience, Medical University of South Carolina, Charleston, SC, United States
- 2Department of Pharmacology and Physiology, Faculty of Medicine, Université de Montréal, Montreal, QC, Canada
- 3Department of Psychiatry, Charité University Medicine Berlin, Campus Charité Mitte, Berlin, Germany
Antipsychotic treatment resistance in schizophrenia remains a major issue in psychiatry. Nearly 30% of patients with schizophrenia do not respond to antipsychotic treatment, yet the underlying neurobiological causes are unknown. All effective antipsychotic medications are thought to achieve their efficacy by targeting the dopaminergic system. Here we review early literature describing the fundamental mechanisms of antipsychotic drug efficacy, highlighting mechanistic concepts that have persisted over time. We then reconsider the original framework for understanding antipsychotic efficacy in light of recent advances in our scientific understanding of the dopaminergic effects of antipsychotics. Based on these new insights, we describe a role for the dopamine transporter in the genesis of both antipsychotic therapeutic response and primary resistance. We believe that this discussion will help delineate the dopaminergic nature of antipsychotic treatment-resistant schizophrenia.
Also Check: How Many Panic Attacks In A Day
Challenges In Diagnosing Schizophrenia
Psychiatric symptoms exist on continua from normal to pathological, meaning the threshold for diagnosis of schizophrenia in clinical practice can be challenging. The clinical diagnosis of schizophrenia relies heavily on the positive symptoms associated with a prolonged psychotic episode. However, a relatively high percentage of the general population report delusional experiences or hallucinations in their lifetime,,, but for most people these are transient. Psychotic symptoms are also not specific to a particular mental disorder. The clinical efficacy of antipsychotic drugs is heavily correlated with their ability to block subcortical dopamine D2 receptors, , suggesting dopamine signalling is important. In spite of this, no consistent relationship between D2 receptors and the pathophysiology of schizophrenia has emerged, . In contrast, the clinical evidence points towards presynaptic dopamine dysfunction as a mediator of psychosis in schizophrenia.