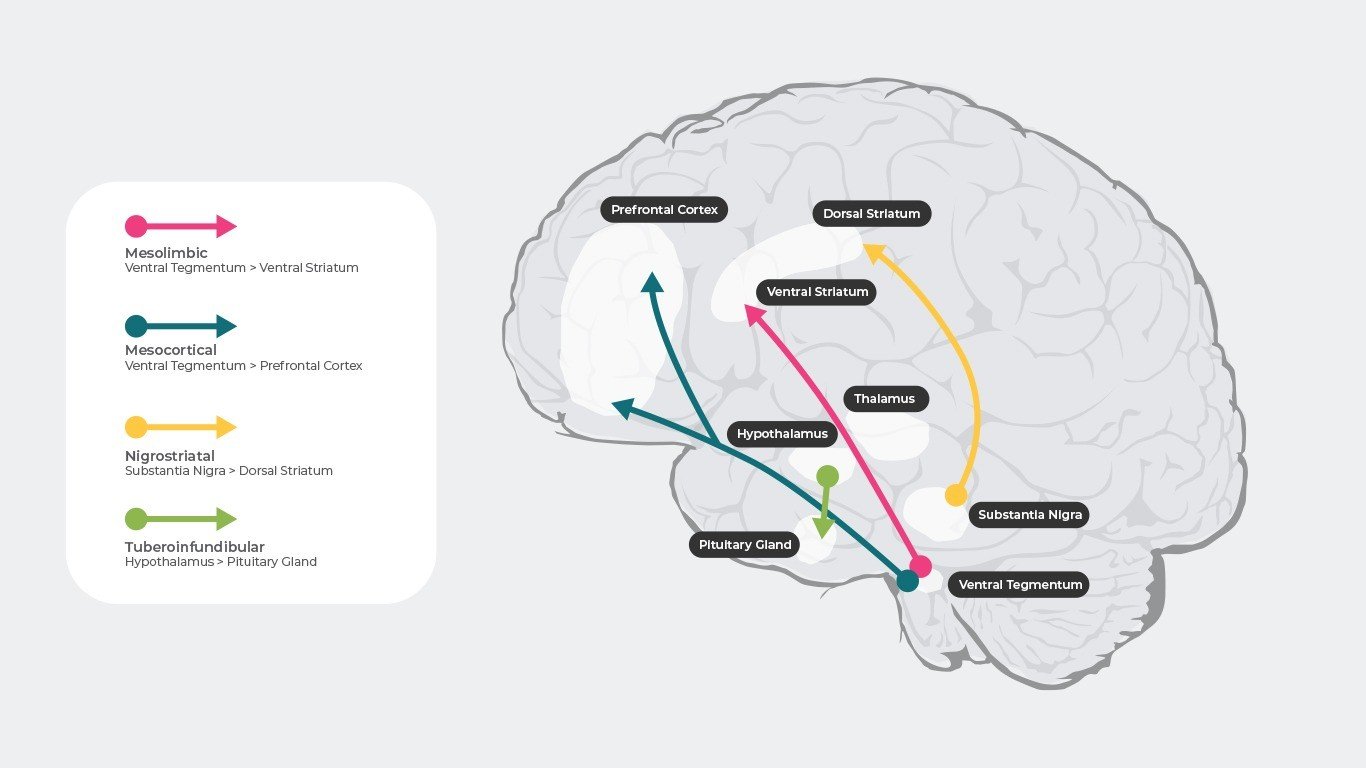
When Neurotransmitters Do Not Work Right
As with many of the body’s processes, things can sometimes go awry. It is perhaps not surprising that a system as vast and complex as the human nervous system would be susceptible to problems.
A few of the things that might go wrong include:
- Neurons might not manufacture enough of a particular neurotransmitter
- Neurotransmitters may be reabsorbed too quickly
- Too many neurotransmitters may be deactivated by enzymes
- Too much of a particular neurotransmitter may be released
When neurotransmitters are affected by disease or drugs, there can be a number of different adverse effects on the body. Diseases such as Alzheimer’s, epilepsy, and Parkinson’s are associated with deficits in certain neurotransmitters.
Health professionals recognize the role that neurotransmitters can play in mental health conditions, which is why medications that influence the actions of the body’s chemical messengers are often prescribed to help treat a variety of psychiatric conditions.
For example, dopamine is associated with such things as addiction and schizophrenia. Serotonin plays a role in mood disorders including depression and OCD. Drugs, such as SSRIs, may be prescribed by physicians and psychiatrists to help treat symptoms of depression or anxiety.
Medications are sometimes used alone, but they may also be used in conjunction with other therapeutic treatments including cognitive-behavioral therapy.
What Neurotransmitters Are Involved In Schizophrenia
DOPAMINE
Two brain chemicals may interact to contribute to the development of psychotic disorders such as schizophrenia, according to a new study. The results suggest abnormal levels of the neurotransmitter glutamate may lead to changes in the levels of another neurotransmitter, dopamine, causing the transition into psychosis.
Beside above, what is the chemical imbalance in schizophrenia? Chemistry: Scientists believe that people with schizophrenia have an imbalance of the brain chemicals or neurotransmitters: dopamine, glutamate and serotonin. These neurotransmitters allow nerve cells in the brain to send messages to each other.
In respect to this, what is the role of dopamine in schizophrenia?
Dopamine gets a lot of attention in brain research because its been linked to addiction. It also plays a role in other psychiatric and movement disorders, like Parkinsons disease. In schizophrenia, dopamine is tied to hallucinations and delusions.
What does serotonin do in schizophrenia?
Moreover, serotonin has been implicated in a variety of behaviors and somatic functions that are disturbed in schizophrenia .
Brain Chemicals And Depression
Researchers have suggested that for some people, having too little of certain substances in the brain could contribute to depression. Restoring the balance of brain chemicals could help alleviate symptomswhich is where the different classes of antidepressant medications may come in.
Even with the help of medications that balance specific neurotransmitters in the brain, depression is a highly complex condition to treat. What proves to be an effective treatment for one person with depression may not work for someone else. Even something that has worked well for someone in the past may become less effective over time, or even stop working, for reasons researchers are still trying to understand.
Researchers continue to try to understand the mechanisms of depression, including brain chemicals, in hopes of finding explanations for these complexities and developing more effective treatments. Depression is a multi-faceted condition, but having an awareness of brain chemistry can be useful for medical and mental health professionals, researchers, and many people who have depression.
Depression Discussion Guide
Don’t Miss: Schizophrenia Physiology
Balance Your Blood Sugar And Avoid Stimulants
Your intake of sugar, refined carbohydrates, caffeine, alcohol and cigarettes, as well as stimulant drugs, all affect the ability to keep ones blood sugar level balanced. On top of this common antipsychotic medication may also further disturb blood sugar control. Stimulant drugs, from amphetamines to cocaine, can induce schizophrenia. The incidence of blood sugar problems and diabetes is also much higher in those with schizophrenia.
Therefore it is strongly advisable to reduce, as much as possible, your intake of sugar, refined carbohydrates, caffeine and stimulant drugs and eat a low glycemic load diet.
Examples Of Important Neurotransmitter Actions

As explained above, the only direct action of a neurotransmitter is to activate a receptor. Therefore, the effects of a neurotransmitter system depend on the connections of the neurons that use the transmitter, and the chemical properties of the receptors that the transmitter binds to.
Here are a few examples of important neurotransmitter actions:
Dont Miss: Does Pristiq Help With Anxiety
Read Also: Definition Of Phobia
Sleep And Schizophrenia Are Intimately Linked
Since sleep regulation involves many neurotransmitter systems and brain circuits, it is likely that the mechanisms generating normal sleep overlap with those that maintain mental health. This would explain why disturbed sleep and schizophrenia are so intimately linked. This was the essence of the seminar given at the Lundbeck Institute on 17th April 2015 by Russell Foster, director of the Sleep and Circadian Neuroscience Unit at the University of Oxford, UK.
Sleep disturbance is common in serious mental illness, and schizophrenia is no exception. It is widely accepted that schizophrenia disrupts sleep and circadian rhythms. But it also seems that disturbances of sleep can precede severe mental illness and may even help cause it.1
Recommended Reading: Does Pristiq Help With Anxiety
Drugs That Influence Neurotransmitters
Perhaps the greatest practical application for the discovery and detailed understanding of how neurotransmitters function has been the development of drugs that impact chemical transmission. These drugs are capable of changing the effects of neurotransmitters, which can alleviate the symptoms of some diseases.
- Agonists vs Antagonists: Some drugs are known as agonists and function by increasing the effects of specific neurotransmitters. Other drugs and referred to as antagonists and act to block the effects of neurotransmission.
- Direct vs Indirect Effects:These neuro-acting drugs can be further broken down based on whether they have a direct or indirect effect. Those that have a direct effect work by mimicking the neurotransmitters because they are very similar in chemical structure. Those that have an indirect impact work by acting on the synaptic receptors.
Drugs that can influence neurotransmission include medications used to treat illness including depression and anxiety, such as SSRIs, tricyclic antidepressants, and benzodiazepines.
Illicit drugs such as heroin, cocaine, and marijuana also have an effect on neurotransmission. Heroin acts as a direct-acting agonist, mimicking the brain’s natural opioids enough to stimulate their associated receptors. Cocaine is an example of an indirect-acting drug that influences the transmission of dopamine.
Recommended Reading: What Is The Meaning Of Phobia
Outstanding Questions And Future Directions
There are a number of outstanding issues when it comes to understanding the role of dopamine and glutamate in schizophrenia. In the case of glutamate, it is not possible to separate extra- and intracellular compartments using MRS, and we cannot accurately probe receptors and synaptic glutamate levels in vivo. As a result, it is not currently possible to precisely characterize the nature of glutamate dysfunction in schizophrenia. It remains unclear whether synaptic glutamate levels are abnormal, whether receptors are altered, and where any alterations might be localized within the brain.
Because of this, it is not clear whether treatments should aim to reduce synaptic glutamate levels or augment glutamatergic neurotransmission. This likely contributes to the fact that to date no glutamate modulating agents exist that demonstrate unequivocal efficacy in schizophrenia.
There is a need for radioligands with reliable binding at the NMDA receptor to allow for investigation of receptor abnormalities in schizophrenia. PET ligands for other proteins involved in glutamatergic signalling, such as AMPA receptors, enzymes involved in glutamate synthesis and metabolism, and the kynurenine pathway, would also represent a considerable advance. In the meantime, other methods, such as functional MRS, 11C-MRS, GluCEST and 7T 1H-MRS, may advance our understanding by allowing more precise inferences regarding the nature of glutamatergic abnormalities in schizophrenia.
Studies On The Dopaminergic Systems
The findings that antipsychotic drugs are dopamine D2 receptor antagonists and that dopamine receptor agonists can cause or exacerbate psychoses has underpinned the long standing dopamine hypothesis of schizophrenia. This hypothesis proposes that overactive dopaminergic pathways in the CNS are central to the pathology of the illness. Recent work on dopaminergic systems, using postmortem tissue, has mainly focused on levels of mRNA for the different dopamine receptors in the cortex of subjects with schizophrenia. Thus, one study reported an increase in mRNA for the dopamine D2longer receptor in the frontal cortex of subjects with schizophrenia. This finding, along with the report of an increase in mRNA for the dopamine D4 receptor in the cortex but not caudate from subjects with schizophrenia, would suggest that there may be abnormalities in the expression of cortical dopamine receptors associated with the illness. Unfortunately, the lack of specific radioligands for the dopamine D4 and D2longer receptors means that it is not possible to determine if these changes in levels of expression have resulted in changes in receptor protein in the cortex from subjects with schizophrenia. This is important as there appears to be no change in the density of global dopamine D2-like or dopamine D1-like receptors in the frontal cortex from subjects with schizophrenia.
You May Like: What Are The Three Stages Of Schizophrenia
Alternate Neurochemical Models In Schizophrenia And Their Interactions With Dopamine
Deviations in dopamine and glutamate have been reported in the prefrontal cortex of schizophrenia patients . NMDA-receptors are involved in releasing dopamine into the striatum and frontal cortex in schizophrenia patients and in rats in an animal model of schizophrenia . These interactions are accompanied by calcium-dependent changes and exchanges between DAT and G72 in various brain regions . In contrast to dopamine receptors, glutamate receptors are found in the subcortical and cortical brain regions . The activity of dopamine is regulated by GABA and glutamate. For example, corticostriatal glutamatergic pathways interact with dopamine terminals , and specific glutamate receptors in the striatum, such as mGlu2, are sensitive to dopamine . High glutamate levels have been found in the dorsal caudate nucleus of schizophrenia patients . Adenosine interacts with glutamate, NMDA-receptors, and dopamine . It can be summarized that NMDA-receptors and D -receptors in cortical brain areas such as the prefrontal cortex and an excess of D -receptors in subcortical brain areas such as the striatum are interconnected with each other through a positive feedback mechanism . However, through its presynaptic action, dopamine reduces the release of glutamate in the pyramidal neurons of layer V in the prefrontal cortex . Dopamine dysregulation in the basal ganglia of schizophrenia patients is an important intrinsic feature in the pathology of schizophrenia and not a medication side effect .
Other Neurotransmitters In Schizophrenia
Clozapine, which is the most effective medication available for treating the symptoms of Schizophrenia, is a very weak blocker of the D2 dopamine receptors. This suggests that there must be other neurotransmitter systems that are also involved in causing Schizophrenia, though so far not much is known about what are these other factors.
One neurotransmitter that has gotten increased attention in recent years for its role in Schizophrenia in glutamate, which targets NMDA receptors in the brain. Phencyclidine and Ketamine are two drugs that block the actions of glutamate at the NMDA receptor, and these drugs can cause both the positive and negative symptoms of Schizophrenia in people who do not have the condition. Increasing the actions of glutamate by using high doses of precursor molecules that are metabolized into glutamate, such as D-serine, glycine and D-cycloserine, show promise in helping to improve the negative symptoms of Schizophrenia.
Read Also: What Is The Meaning Of Phobia
Stay Informed On Keep The Body In Mind
Continue to receive information on the pathophysiology of and comorbidities associated with bipolar disorder or schizophrenia from KEEP THE BODY IN MIND.
References:1.J Psychosom Res.2.Schizophr Res.3.Psychiatr Serv.4.Lancet HIV.5.J Clin Psychiatry.6.Nicotine Tob Res.7.Front Psychiatry.8.J Nerv Ment Dis.9.Neurosci Biobehav Rev.10.Eur Psychiatry.11.Front Psychiatry.12.Curr Psychiatry Rep.13.Int J Mol Sci.14.Adv Pharmacol.15.Mol Psychiatry.16.Front Neurosci.17.Eur J Endocrinol.18.J Nutr Metab.19.Diabetes.20.Can J Psychiatry.21.J Psychopharmacol.22.Mol Psychiatry.23.Schizophr Res.
We have updated our Privacy Policy. Please review our Privacy Policy. This website uses cookies. By using our website without changing your cookie settings you agree to our use of cookies as described in our Privacy Policy.
This website contains information intended for U.S. healthcare professionals only.
A Scientific Team Has Shown That The Release Of Neurotransmitters In The Brain Is Impaired In Patients With Schizophrenia Who Have A Rare Single
Significantly, the results from the research with human-derived neurons validated previous and new experiments that found the same major decrease in neurotransmitter release and synaptic signaling in genetically engineered human neurons with the same genetic variant the deletion of neurexin 1 . NRXN1 is a protein-coding gene at the synapse, a cellular junction that connects two nerve cells to communicate efficiently.
Both the research with human-derived and engineered human neurons also found an increase in the levels of CASK, an NRXN1-binding protein, which were associated with changes in gene expression.
Losing one copy of this neurexin 1 gene somehow contributes to the etiology or the disease mechanism in these schizophrenia patients, says molecular neuroscientist ChangHui Pak, assistant professor of biochemistry and molecular biology at the University of Massachusetts Amherst and lead author of the research published in the Proceedings of the National Academy of Sciences. It causes a deficit in neural communication.
“Although this single-gene mutation puts people at risk for schizophrenia, autism, Tourette syndrome and other neuropsychiatric disorders, at the end of the day, we dont know what causes schizophrenia. This variant gives us insight into what cellular pathways would be perturbed among people with schizophrenia and a lead to study this biology. ChangHui Pak, lead author of the research
Read on
Also Check: Medical Definition Of Phobia
Box : The Syndrome Of Schizophrenia
-
Schizophrenia is likely to be a syndrome and its symptoms could be generated by different pathologies.
-
As is proving the case with Alzheimer’s disease, it is likely that abnormalities in different pathways of the CNS will account for the onset of symptoms in subsets of individuals with schizophrenia.
Clearly further study is warranted to determine the extent of changes in the Wnt pathway in postmortem CNS from subjects with schizophrenia and to understand how this pathway may be involved in the pathological processes leading to the onset of the illness.
In addition to the more established lines of experiments on neurotransmitter release and Wnt pathway there are early reports of changes in lipoproteins, cell guidance proteins such as reelin, and proteins in the apoptotic pathways such as Bcl-2 in the CNS from subjects with schizophrenia. Changes in these key pathways in schizophrenia would be expected to profoundly disrupt CNS functioning. However, there is now a growing body of data that suggests that changes in non-neurotransmitter specific proteins could be important in the underlying pathology of schizophrenia. It is still not clear whether the extensive changes in neurotransmitter associated proteins are simply a consequence of changes in such proteins.
Consequences Of Glutamatergic Dysfunction
Unlike dopamine neurons, which are restricted to relatively well circumscribed anatomical pathways, glutamate signalling occurs ubiquitously throughout the brain and, as a result, dysfunction of this system has the potential to account for a wide range of impairments.
However, given the limitations regarding techniques for directly quantifying the glutamate system in vivo, there is a paucity of direct evidence regarding the precise nature of glutamatergic dysfunction in schizophrenia, and studies looking at the relationship between 1H-MRS measures of glutamate and symptom severity have produced inconsistent findings. Indeed, both increased and decreased level of glutamate as measured by 1H-MRS have been proposed to support a hypothesis of NMDA hypofunction in schizophrenia, .
NMDA receptors, sparse coding and memory
NMDA receptors play a vital role in orchestrating several cog-nitive processes, including working memory. One of the mechanisms involved in the efficient cortical representation of information is that of sparse coding.
Excitatory/inhibitory balance and neuronal oscillations
Synchronized neuronal oscillations are associated with a wide range of cognitive processes, such as working memory. These oscillations result from a tightly maintained balance between excitatory and inhibitory populations of neurons, and can be measured in vivo using electroencephalogram .
Don’t Miss: What Is A Depression On A Map
Indirect Evidence For Glutamates Involvement
Post mortem and genetic studies
Post mortem studies provide evidence for alterations to glutamatergic functioning in schizophrenia. Studies of NMDA receptor expression in post mortem samples have produced some positive findings such as reduced NMDAR1 subunit density in the superior frontal cortex and the superior temporal cortex . However, overall findings regarding NMDA receptor density have been inconsistent . It seems that the abnormality in schizophrenia may primarily be aberrant glutamate receptor localisation as opposed to a generalised deficit . This abnormality could arise as a result of changes in glutamate receptor trafficking molecules . Furthermore there is evidence for a variety of functional changes affecting the intracellular effects of the NMDA receptor that would have a major impact on glutamatergic signalling.
The potential role of glutamate in the pathophysiology of schizophrenia is also supported by recent genetics findings . GRIN2A which codes for an NMDA receptor subunit was found to be associated with schizophrenia, as was SRR which plays a key role in pathways leading to the activation of the NMDA receptor.
NMDA receptor antagonists
Read Also: Prodromal Symptoms Of Schizophrenia Are Evident
Studies On The Glutamatergic Systems
The ability of phencyclidine, a glutamate receptor ion channel blocker, to induce or exacerbate a schizophrenic-like psychoses, has been central to the hypotheses that changed glutamatergic function is involved in the pathology of schizophrenia. This has led to an extensive investigation of glutamatergic markers in postmortem CNS tissue from subjects with schizophrenia. There are two major families of glutamate receptors. One family is a group of ionotropic glutamate receptors made up of the N-methyl-d-aspartate , the -amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid and the kainate receptors. All these receptors are made up of a combination of specific subunits, which assemble in the membrane to form cation conductance channels. The other family of receptors are known as the metabotropic receptors and are G-protein coupled receptors.
In contrast to studies in the thalamus and hippocampus, it has been reported that neither AMPA receptor radioligand binding nor levels of mRNA for AMPA receptor subunits are altered in the frontal cortex of subjects with schizophrenia. However, levels of mRNA for the NR1, gluR1, gluR7, and KA1 subunits of glutamate receptors have been reported as being decreased in the cortex of schizophrenic subjects not receiving antipsychotic drugs within six months of death. Significantly, in this study decreased levels of mRNA for subunits of the glutamate receptors were not observed in subjects who were receiving antipsychotic drugs up until death.
Recommended Reading: Is Tequila A Stimulant Or Depressant